Editors’ Picks: January Highlights from AACR Journals
To kick off the new year, the editors of the 10 AACR journals highlighted two phase I clinical trials, a strategy for increasing the immunogenicity of KRAS-mutated cancers, and insight into the progression of a type of leukemia, among other studies.
Read the abstracts of this month’s featured studies below, and follow the links to read the articles in full. As always, the highlighted articles are freely available for a limited time.
Journal: Blood Cancer Discovery
Extracellular Vesicle Secretion by Leukemia Cells In Vivo Promotes CLL Progression by Hampering Antitumor T-cell Responses
Small extracellular vesicle (sEV, or exosome) communication among cells in the tumor microenvironment has been modeled mainly in cell culture, whereas their relevance in cancer pathogenesis and progression in vivo is less characterized. Here we investigated cancer–microenvironment interactions in vivo using mouse models of chronic lymphocytic leukemia (CLL). sEVs isolated directly from CLL tissue were enriched in specific miRNA and immune-checkpoint ligands. Distinct molecular components of tumor-derived sEVs altered CD8+ T-cell transcriptome, proteome, and metabolome, leading to decreased functions and cell exhaustion ex vivo and in vivo. Using antagomiRs and blocking antibodies, we defined specific cargo-mediated alterations on CD8+ T cells. Abrogating sEV biogenesis by Rab27a/b knockout dramatically delayed CLL pathogenesis. This phenotype was rescued by exogenous leukemic sEV or CD8+ T-cell depletion. Finally, high expression of sEV-related genes correlated with poor outcomes in CLL patients, suggesting sEV profiling as a prognostic tool. In conclusion, sEVs shape the immune microenvironment during CLL progression.
This article is highlighted and featured on the cover of the January issue. A related commentary can be found here.
Journal: Cancer Discovery
Creating MHC-Restricted Neoantigens with Covalent Inhibitors That Can Be Targeted by Immune Therapy
Intracellular oncoproteins can be inhibited with targeted therapy, but responses are not durable. Immune therapies can be curative, but most oncogene-driven tumors are unresponsive to these agents. Fragments of intracellular oncoproteins can act as neoantigens presented by the major histocompatibility complex (MHC), but recognizing minimal differences between oncoproteins and their normal counterparts is challenging.
We have established a platform technology that exploits hapten–peptide conjugates generated by covalent inhibitors to create distinct neoantigens that selectively mark cancer cells. Using the FDA-approved covalent inhibitors sotorasib and osimertinib, we developed “HapImmune” antibodies that bind to drug–peptide conjugate/MHC complexes but not to the free drugs. A HapImmune-based bispecific T-cell engager selectively and potently kills sotorasib-resistant lung cancer cells upon sotorasib treatment. Notably, it is effective against KRASG12C-mutant cells with different HLA supertypes, HLA-A*02 and A*03/11, suggesting loosening of MHC restriction. Our strategy creates targetable neoantigens by design, unifying targeted and immune therapies.
This article is highlighted and featured on the cover of the January issue. A related commentary can be found here.
Journal: Cancer Epidemiology, Biomarkers & Prevention
Impact and Recovery from COVID-19–Related Disruptions in Colorectal Cancer Screening and Care in the US: A Scenario Analysis
Background: Many colorectal cancer–related procedures were suspended during the COVID-19 pandemic. In this study, we predict the impact of resulting delays in screening (colonoscopy, FIT, and sigmoidoscopy) and diagnosis on colorectal cancer–related outcomes, and compare different recovery scenarios. Methods: Using the MISCAN-Colon model, we simulated the US population and evaluated different impact and recovery scenarios. Scenarios were defined by the duration and severity of the disruption (percentage of eligible adults affected), the length of delays, and the duration of the recovery. During recovery (6, 12 or 24 months), capacity was increased to catch up missed procedures. Primary outcomes were excess colorectal cancer cases and –related deaths, and additional colonoscopies required during recovery. Results: With a 24-month recovery, the model predicted that the US population would develop 7,210 (0.18%) excess colorectal cancer cases during 2020–2040, and 6,950 (0.65%) excess colorectal cancer–related deaths, and require 108,500 (8.6%) additional colonoscopies per recovery month, compared with a no-disruption scenario. Shorter recovery periods of 6 and 12 months, respectively, decreased excess colorectal cancer–related deaths to 4,190 (0.39%) and 4,580 (0.43%), at the expense of 260,200–590,100 (20.7%–47.0%) additional colonoscopies per month. Conclusions: The COVID-19 pandemic will likely cause more than 4,000 excess colorectal cancer–related deaths in the US, which could increase to more than 7,000 if recovery periods are longer. Impact: Our results highlight that catching-up colorectal cancer–related services within 12 months provides a good balance between required resources and mitigation of the impact of the disruption on colorectal cancer–related deaths.
This article is highlighted in the January issue.
Journal: Cancer Immunology Research
The Conventional Dendritic Cell 1 Subset Primes CD8+ T Cells and Traffics Tumor Antigen to Drive Antitumor Immunity in the Brain
The central nervous system (CNS) antigen-presenting cell (APC) that primes antitumor CD8+ T-cell responses remains undefined. Elsewhere in the body, the conventional dendritic cell 1 (cDC1) performs this role. However, steady-state brain parenchyma cDC1 are extremely rare; cDCs localize to the choroid plexus and dura. Thus, whether the cDC1 play a function in presenting antigen derived from parenchymal sources in the tumor setting remains unknown. Using preclinical glioblastoma (GBM) models and cDC1-deficient mice, we explored the presently unknown role of cDC1 in CNS antitumor immunity. We determined that, in addition to infiltrating the brain tumor parenchyma itself, cDC1 prime neoantigen-specific CD8+ T cells against brain tumors and mediate checkpoint blockade-induced survival benefit. We observed that cDC, including cDC1, isolated from the tumor, the dura, and the CNS-draining cervical lymph nodes harbored a traceable fluorescent tumor antigen. In patient samples, we observed several APC subsets (including the CD141+ cDC1 equivalent) infiltrating glioblastomas, meningiomas, and dura. In these same APC subsets, we identified a tumor-specific fluorescent metabolite of 5-aminolevulinic acid, which fluorescently labeled tumor cells during fluorescence-guided GBM resection. Together, these data elucidate the specialized behavior of cDC1 and suggest that cDC1 play a significant role in CNS antitumor immunity.
This article is featured on the cover of the January issue.
Journal: Cancer Prevention Research
Phase I Dose Escalation Study of Topical Bexarotene in Women at High Risk for Breast Cancer
Agents that can reduce the incidence of hormone receptor negative breast cancer are currently lacking. Rexinoids such as bexarotene significantly reduced mammary tumor development in preclinical mouse models. Oral bexarotene in BRCA mutation carriers significantly decreased cyclin D1 in breast cells, suggesting biological activity on breast tissue. This study evaluated topical bexarotene 1% gel applied to one unaffected breast in women at high risk for breast cancer for 4 weeks to assess safety and toxicity. Secondary objectives included assessment of bexarotene concentrations in the plasma and breast tissue. In the dose escalation phase, women were assigned to one of three different dose levels: 10 mg (1 mL) every other day, 10 mg (1 mL) daily, 20 mg (2 mL) daily. Dose-limiting toxicity (DLT) was defined as a grade 2 skin adverse event for at least 6 days or any grade 3 or 4 adverse event related to study drug. A total of 14 women were enrolled with 10 participants at the every other day dose level and 4 participants at daily dosing. Two skin DLTs were experienced at daily dosing and therefore further enrollment was discontinued per protocol. An additional 10 participants were enrolled at the MTD as part of the dose expansion phase. These individuals tolerated the treatment with minimal adverse events. Maculopapular rash at the treatment site was the most common adverse event related to study drug and resolved within a few days of discontinuation. Bexarotene was detectable in breast tissue at the 10 mg daily every other day dose.
Journal: Cancer Research (January 1 issue)
Spatial Transcriptomic Analysis of a Diverse Patient Cohort Reveals a Conserved Architecture in Triple-Negative Breast Cancer
Triple-negative breast cancer (TNBC) is an aggressive disease that disproportionately affects African American (AA) women. Limited targeted therapeutic options exist for patients with TNBC. Here, we employ spatial transcriptomics to interrogate tissue from a racially diverse TNBC cohort to comprehensively annotate the transcriptional states of spatially resolved cellular populations. A total of 38,706 spatial features from a cohort of 28 sections from 14 patients were analyzed. Intratumoral analysis of spatial features from individual sections revealed heterogeneous transcriptional substructures. However, integrated analysis of all samples resulted in nine transcriptionally distinct clusters that mapped across all individual sections. Furthermore, novel use of join count analysis demonstrated nonrandom directional spatial dependencies of the transcriptionally defined shared clusters, supporting a conserved spatio-transcriptional architecture in TNBC. These findings were substantiated in an independent validation cohort comprising 17,861 spatial features representing 15 samples from 8 patients. Stratification of samples by race revealed race-associated differences in hypoxic tumor content and regions of immune-rich infiltrate. Overall, this study combined spatial and functional molecular analyses to define the tumor architecture of TNBC, with potential implications in understanding TNBC disparities.
This article is featured on the cover of the January issue.
Journal: Cancer Research (January 15 issue)
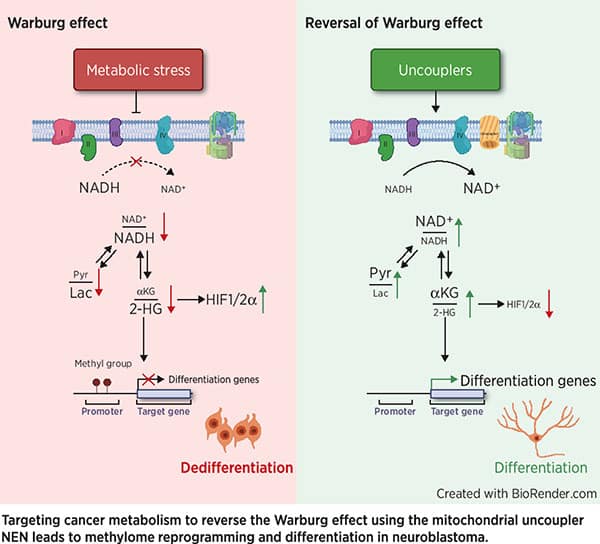
Mitochondrial Uncoupling Induces Epigenome Remodeling and Promotes Differentiation in Neuroblastoma
The Warburg effect is the major metabolic hallmark of cancer. According to Warburg himself, the consequence of the Warburg effect is cell dedifferentiation. Therefore, reversing the Warburg effect might be an approach to restore cell differentiation in cancer. In this study, we used a mitochondrial uncoupler, niclosamide ethanolamine (NEN), to activate mitochondrial respiration, which induced neural differentiation in neuroblastoma cells. NEN treatment increased the NAD+/NADH and pyruvate/lactate ratios and also the α-ketoglutarate/2-hydroxyglutarate (2-HG) ratio. Consequently, NEN treatment induced promoter CpG island demethylation and epigenetic landscape remodeling, activating the neural differentiation program. In addition, NEN treatment upregulated p53 but downregulated N-Myc and β-catenin signaling in neuroblastoma cells. Importantly, even under hypoxia, NEN treatment remained effective in inhibiting 2-HG generation, promoting DNA demethylation, and suppressing hypoxia-inducible factor signaling. Dietary NEN intervention reduced tumor growth rate, 2-HG levels, and expression of N-Myc and β-catenin in tumors in an orthotopic neuroblastoma mouse model. Integrative analysis indicated that NEN treatment upregulated favorable prognosis genes and downregulated unfavorable prognosis genes, which were defined using multiple neuroblastoma patient datasets. Altogether, these results suggest that mitochondrial uncoupling is an effective metabolic and epigenetic therapy for reversing the Warburg effect and inducing differentiation in neuroblastoma. A related commentary can be found here.
Journal: Clinical Cancer Research (January 1 issue)
Pilot Study of ONCOS-102 and Pembrolizumab: Remodeling of the Tumor Microenvironment and Clinical Outcomes in Anti–PD-1–Resistant Advanced Melanoma
Purpose: Intratumoral oncolytic virotherapy may overcome anti–PD(L)-1 resistance by triggering pro-inflammatory remodeling of the tumor microenvironment. This pilot study investigated ONCOS-102 (oncolytic adenovirus expressing GM-CSF) plus anti–programmed cell death protein 1 (PD)-1 therapy in anti–PD-1–resistant melanoma. Patients and Methods: Patients with advanced melanoma progressing after prior PD-1 blockade received intratumoral ONCOS-102 either as priming with 3 doses (3 × 1011 viral particles) during Week 1 [Part 1 (sequential treatment)] or as 4-dose priming and 8 booster doses every 3 weeks [Part 2 (combination treatment)]. From Week 3, all patients received pembrolizumab every 3 weeks (≤8 doses). The primary endpoint was safety. Objective response rate (ORR), progression-free survival, and immunologic activation in repeat biopsies were also investigated. Results: In 21 patients (Part 1, n = 9; Part 2, n = 12) ONCOS-102 plus pembrolizumab was well tolerated: most adverse events (AE) were mild/moderate in severity. Pyrexia (43%), chills (43%), and nausea (28%) were the most common ONCOS-102–related AEs. There were no dose-limiting toxicities. ORR was 35% [response evaluation in solid tumors (RECIST) 1.1, irRECIST]. Reduction in size of ≥1 non-injected lesions observed in 53% patients indicated a systemic effect. In injected tumors, persistent immune-related gene expression and T-cell infiltration were associated with clinical benefit. Viral persistence and efficacy in injected and non-injected lesions without additional toxicity supported Part 2 dosing regimen in future studies. Conclusions: ONCOS-102 plus pembrolizumab was well tolerated and led to objective responses in patients with anti–PD-1–resistant advanced melanoma. ONCOS-102 promoted T-cell infiltration, particularly cytotoxic CD8+ T cells, which persisted at Week 9, driving clinical benefit. Further investigation of ONCOS-102 plus PD-1 blockade is warranted.
This article is highlighted in the January 1st issue. A related commentary can be found here.
Journal: Clinical Cancer Research (January 15 issue)
Intraperitoneal Monocytes plus IFNs as a Novel Cellular Immunotherapy for Ovarian Cancer: Mechanistic Characterization and Results from a Phase I Clinical Trial
Purpose: Ovarian cancer is the most lethal gynecologic cancer and intrinsically resistant to checkpoint immunotherapies. We sought to augment innate immunity, building on previous work with IFNs and monocytes. Patients and Methods: Preclinical experiments were designed to define the mechanisms of cancer cell death mediated by the combination of IFNs α and γ with monocytes. We translated these preclinical findings into a phase I trial of autologous IFN-activated monocytes administered intraperitoneally to platinum-resistant or -refractory ovarian cancer patients. Results: IFN-treated monocytes induced caspase 8–dependent apoptosis by the proapoptotic TRAIL and mediated by the death receptors 4 and 5 (DR4 and DR5, respectively) on cancer cells. Therapy was well tolerated with evidence of clinical activity, as 2 of 9 evaluable patients had a partial response by RECIST criteria, and 1 additional patient had a CA-125 response. Upregulation of monocyte-produced TRAIL and cytokines was confirmed in peripheral blood. Long-term responders had alterations in innate and adaptive immune compartments. Conclusions: Given the mechanism of cancer cell death, and the acceptable tolerability of the clinical regimen, this platform presents a possibility for future combination therapies to augment anticancer immunity.
This article is highlighted in the January 15th issue. A related commentary can be found here.
Journal: Molecular Cancer Research
Mitochondrial Aconitase ACO2 Links Iron Homeostasis with Tumorigenicity in Non–Small Cell Lung Cancer
The ability of a patient tumor to engraft an immunodeficient mouse is the strongest known independent indicator of poor prognosis in early-stage non–small cell lung cancer (NSCLC). Analysis of primary NSCLC proteomes revealed low-level expression of mitochondrial aconitase (ACO2) in the more aggressive, engrafting tumors. Knockdown of ACO2 protein expression transformed immortalized lung epithelial cells, whereas upregulation of ACO2 in transformed NSCLC cells inhibited cell proliferation in vitro and tumor growth in vivo. High level ACO2 increased iron response element binding protein 1 (IRP1) and the intracellular labile iron pool. Impaired cellular proliferation associated with high level ACO2 was reversed by treatment of cells with an iron chelator, whereas increased cell proliferation associated with low level ACO2 was suppressed by treatment of cells with iron. Expression of CDGSH iron-sulfur (FeS) domain-containing protein 1 [CISD1; also known as mitoNEET (mNT)] was modulated by ACO2 expression level and inhibition of mNT by RNA interference or by treatment of cells with pioglitazone also increased iron and cell death. Hence, ACO2 is identified as a regulator of iron homeostasis and mNT is implicated as a target in aggressive NSCLC.
This article is highlighted in the January issue.
Journal: Molecular Cancer Therapeutics
Antibody–Drug Conjugate Sacituzumab Govitecan Drives Efficient Tissue Penetration and Rapid Intracellular Drug Release
Antibody–drug conjugates (ADC) are a rapidly growing class of targeted cancer treatments, but the field has experienced significant challenges from their complex design. This study examined the multiscale distribution of sacituzumab govitecan (SG; Trodelvy), a recently clinically approved ADC, to clarify the mechanism(s) of efficacy given its unique design strategy. We employed a multiscale quantitative pharmacokinetic approach, including near-infrared fluorescence imaging, single-cell flow cytometry measurements, payload distribution via γH2AX pharmacodynamic staining, and a novel dual-labeled fluorescent technique to track the ADC and payload in a high trophoblast cell-surface antigen 2 expression xenograft model of gastric cancer (NCI-N87). We found that rapid release of the SN-38 payload from the hydrolysable linker inside cells imparts more DNA damage in vitro and in vivo than an ADC with a more stable enzyme cleavable linker. With SG, little to no extracellular payload release in the tumor was observed using a dual-labeled fluorescence technique, although bystander effects were detected. The high dosing regimen allowed the clinical dose to reach the majority of cancer cells, which has been linked to improved efficacy. In addition, the impact of multiple doses (day 1 and day 8) of a 21-day cycle was found to further improve tissue penetration despite not changing tumor uptake [percent injected dose per gram (%ID/g)] of the ADC. These results show increased ADC efficacy with SG can be attributed to efficient tumor penetration and intracellular linker cleavage after ADC internalization. This quantitative approach to study multiscale delivery can be used to inform the design of next-generation ADCs and prodrugs for other targets.
Journal: Cancer Research Communications
Complex Patterns of Genomic Heterogeneity Identified in 42 Tumor Samples and ctDNA of a Pulmonary Atypical Carcinoid Patient
Tumor evolution underlies many challenges facing precision oncology, and improving our understanding has the potential to improve clinical care. This study represents a rare opportunity to study tumor heterogeneity and evolution in a patient with an understudied cancer type. A patient with pulmonary atypical carcinoid, a neuroendocrine tumor, metastatic to 90 sites, requested and consented to donate tissues for research. 42 tumor samples collected at rapid autopsy from 14 anatomically distinct sites were analyzed through DNA whole-exome sequencing and RNA sequencing, and five analyzed through linked-read sequencing. Targeted DNA sequencing was completed on two clinical tissue biopsies and one blood plasma sample. Chromosomal alterations and gene variants accumulated over time, and specific chromosomal alterations preceded the single predicted gene driver variant (ARID1A). At the time of autopsy, all sites shared the gain of one copy of Chr 5, loss of one copy of Chr 6 and 21, chromothripsis of one copy of Chr 11, and 39 small variants. Two tumor clones (carrying additional variants) were detected at metastatic sites, and occasionally in different regions of the same organ (e.g., within the pancreas). Circulating tumor DNA (ctDNA) sequencing detected shared tumor variants in the blood plasma and captured marked genomic heterogeneity, including all metastatic clones but few private tumor variants. This study describes genomic tumor evolution and dissemination of a pulmonary atypical carcinoid donated by a single generous patient. It highlights the critical role of chromosomal alterations in tumor initiation and explores the potential of ctDNA analysis to represent genomically heterogeneous disease.
Source link
#Editors #Picks #January #Highlights #AACR #Journals