January 2024 Highlights from the AACR Journals

If the winter weather has you feeling chilly, cozy up with the January 2024 edition of Editors’ Picks—selected articles from the editors of the 10 AACR journals. This month, we bring you a study of disparities in genetic testing and precision medicine; a look at unique features of the gut microbiome in patients with early-onset colorectal cancer; an updated analysis from the monarcHER clinical trial; and more.
The abstracts of the selected studies are included below, and each article is freely available for a limited time.
Journal: Blood Cancer Discovery
Multiple myeloma (MM) is a malignancy that is often driven by MYC and that is sustained by IRF4, which are upregulated by super-enhancers. IKZF1 and IKZF3 bind to super-enhancers and can be degraded using immunomodulatory imide drugs (IMiD). Successful IMiD responses downregulate MYC and IRF4; however, this fails in IMiD-resistant cells. MYC and IRF4 downregulation can also be achieved in IMiD-resistant tumors using inhibitors of BET and EP300 transcriptional coactivator proteins; however, in vivo these drugs have a narrow therapeutic window. By combining IMiDs with EP300 inhibition, we demonstrate greater downregulation of MYC and IRF4, synergistic killing of myeloma in vitro and in vivo, and an increased therapeutic window. Interestingly, this potent combination failed where MYC and IRF4 expression was maintained by high levels of the AP-1 factor BATF. Our results identify an effective drug combination and a previously unrecognized mechanism of IMiD resistance.
Significance: These results highlight the dependence of MM on IKZF1-bound super-enhancers, which can be effectively targeted by a potent therapeutic combination pairing IMiD-mediated degradation of IKZF1 and IKZF3 with EP300 inhibition. They also identify AP-1 factors as an unrecognized mechanism of IMiD resistance in MM.
This article was highlighted and featured on the cover of the January issue, and a related commentary is available here.
Journal: Cancer Discovery
GTP Signaling Links Metabolism, DNA Repair, and Responses to Genotoxic Stress
How cell metabolism regulates DNA repair is incompletely understood. Here, we define a GTP-mediated signaling cascade that links metabolism to DNA repair and has significant therapeutic implications. GTP, but not other nucleotides, regulates the activity of Rac1, a guanine nucleotide-binding protein, which promotes the dephosphorylation of serine 323 on Abl-interactor 1 (Abi-1) by protein phosphatase 5 (PP5). Dephosphorylated Abi-1, a protein previously not known to activate DNA repair, promotes nonhomologous end joining. In patients and mouse models of glioblastoma, Rac1 and dephosphorylated Abi-1 mediate DNA repair and resistance to standard-of-care genotoxic treatments. The GTP–Rac1–PP5–Abi-1 signaling axis is not limited to brain cancer, as GTP supplementation promotes DNA repair and Abi-1-S323 dephosphorylation in nonmalignant cells and protects mouse tissues from genotoxic insult. This unexpected ability of GTP to regulate DNA repair independently of deoxynucleotide pools has important implications for normal physiology and cancer treatment.
Significance: A newly described GTP-dependent signaling axis is an unexpected link between nucleotide metabolism and DNA repair. Disrupting this pathway can overcome cancer resistance to genotoxic therapy while augmenting it can mitigate genotoxic injury of normal tissues.
This article was highlighted in the January issue.
Journal: Cancer Epidemiology, Biomarkers & Prevention
Racial Disparities in Accessing Care along the Continuum of Cancer Genetic Service Delivery
Background: Public health calls to ensure equity in genomics and precision medicine necessitate a closer examination of how these efforts might differentially affect access to genetic services across demographic subgroups. This study set out to examine racial/ethnic disparities along the cancer genetic service delivery continuum.
Methods: Retrospective data are drawn from 15 clinical sites across 6 U.S. States. Individuals who screened at-risk for hereditary cancer were: (i) referred/scheduled to see a genetic counselor (referral workflow), or (ii) offered genetic testing at the point-of-care (POC testing workflow). Logistic regression analyses evaluated the associations between race/ethnicity and several outcomes including appointment scheduling, genetic counseling, and genetic testing, controlling for demographics, clinical factors, and county-level covariates.
Results: A total of 14,527 patients were identified at-risk. Genetic testing uptake was significantly higher at POC sites than referral sites (34% POC vs. 11% referral, P < 0.001). Race/ethnicity was significantly associated with testing uptake among all sites, with non-Hispanic Blacks having lower odds of testing compared with non-Hispanic Whites [aOR = 0.84; 95% confidence interval (CI), 0.71–1.00; P = 0.049]. Moreover, this disparity was observed at referral sites, but not POC sites. Among patients scheduled, non-Hispanic Blacks had lower odds of counseling (aOR = 0.28; 95% CI, 0.17–0.47; P < 0.001).
Conclusions: Findings suggest that factors influencing genetic counseling show rates may be driving disparities in genetic testing.
Impact: Strategies to reduce barriers to seeing a genetic counselor, including modifications to clinical workflow, may help mitigate racial/ethnic disparities in genetic testing.
This article was highlighted in the January issue.
Journal: Cancer Immunology Research
Intracellular K+ Limits T-cell Exhaustion and Preserves Antitumor Function
T cells are often compromised within cancers, allowing disease progression. We previously found that intratumoral elevations in extracellular K+, related to ongoing cell death, constrained CD8+ T-cell Akt–mTOR signaling and effector function. To alleviate K+-mediated T-cell dysfunction, we pursued genetic means to lower intracellular K+. CD8+ T cells robustly and dynamically express the Na+/K+ ATPase, among other K+ transporters. CRISPR-Cas9–mediated disruption of the Atp1a1 locus lowered intracellular K+ and elevated the resting membrane potential (i.e., Vm, Ψ). Despite compromised Ca2+ influx, Atp1a1-deficient T cells harbored tonic hyperactivity in multiple signal transduction cascades, along with a phenotype of exhaustion in mouse and human CD8+ T cells. Provision of exogenous K+ restored intracellular levels in Atp1a1-deficient T cells and prevented damaging levels of reactive oxygen species (ROS), and both antioxidant treatment and exogenous K+ prevented Atp1a1-deficient T-cell exhaustion in vitro. T cells lacking Atp1a1 had compromised persistence and antitumor activity in a syngeneic model of orthotopic murine melanoma. Translational application of these findings will require balancing the beneficial aspects of intracellular K+ with the ROS-dependent nature of T-cell effector function.
A related commentary is available here.
Journal: Cancer Prevention Research
Early-onset Colon Cancer Shows a Distinct Intestinal Microbiome and a Host–Microbe Interaction
The incidence rate of colorectal cancer in younger adults has been rising in developed countries. This trend may be attributed to environmental exposures as a result of lifestyle changes. Many of the lifestyle factors that promote colorectal cancer can also affect the gut microbiome, which may be associated with colorectal cancer risks. The role of the microbiome in the ongoing rise of early-onset colorectal cancer is unknown. Here, we aimed to investigate age-related differences in the gut microbiome of patients with colorectal cancer and healthy individuals by examining both the fecal and tumor microbiomes. We utilized the publicly accessible data on fecal shotgun metagenomics from CuratedMetagenomeData and TCGA via the GDC Data Portal. Comparison of 701 colorectal cancer and 693 controls revealed that microbial features were age dependent, with a significant difference in species enrichment between early-onset (<50 years) and late-onset (>65 years) patients with colorectal cancer. Analysis of the tumor-associated microbiome in a separate dataset of 85 patients with colorectal cancer verified age-specific differences in taxon abundance between early- and late-onset patients with colorectal cancer. Finally, using host gene expression data, we found a stronger microbe–host interaction in early- vs. late-onset colorectal cancers. Altogether, these findings indicate that microbial features were age-dependent with stronger microbial–host interactions at the tumor site in early-onset colorectal cancers, suggesting a direct role of microbes in tumorigenesis via interaction with cancer-related pathways in this age group.
Prevention Relevance: Early-onset colorectal cancer is on the rise, presumably because of changes in environmental exposures. Lifestyle changes may contribute to colorectal cancer via alterations in gut microbes. Here, we show that microbial association with colorectal cancer is age-dependent, and microbe interactions with tumor pathways are stronger in young versus older colorectal cancers.
This article was featured on the cover of the January issue.
Journal: Cancer Research (January 1 issue)
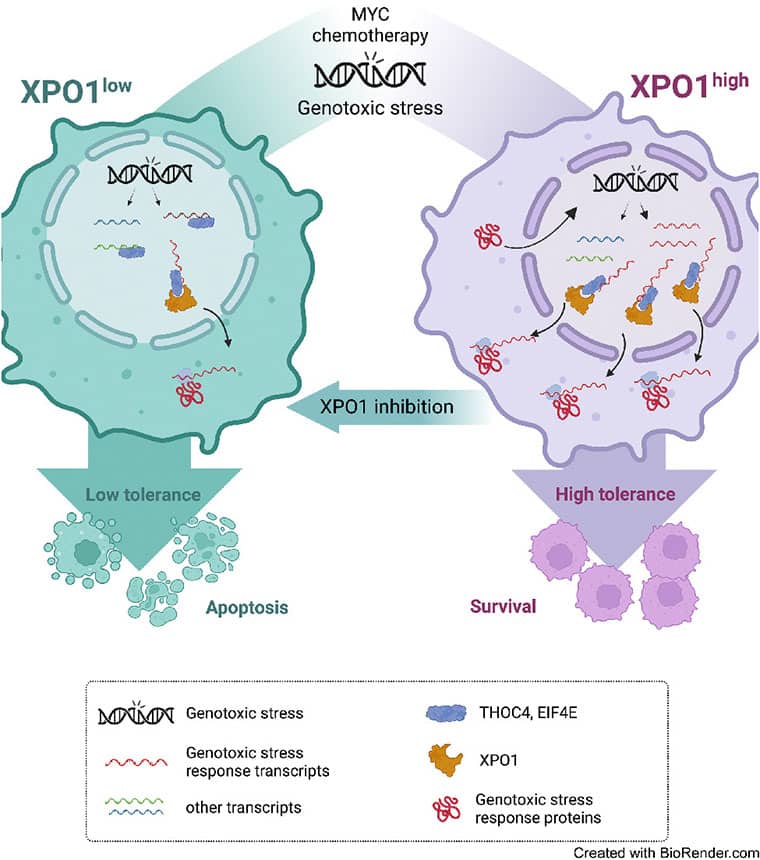
Exportin-1 (XPO1), the main soluble nuclear export receptor in eukaryotic cells, is frequently overexpressed in diffuse large B-cell lymphoma (DLBCL). A selective XPO1 inhibitor, selinexor, received approval as single agent for relapsed or refractory (R/R) DLBCL. Elucidating the mechanisms by which XPO1 overexpression supports cancer cells could facilitate further clinical development of XPO1 inhibitors. We uncovered here that XPO1 overexpression increases tolerance to genotoxic stress, leading to a poor response to chemoimmunotherapy. Upon DNA damage induced by MYC expression or exogenous compounds, XPO1 bound and exported EIF4E and THOC4 carrying DNA damage repair mRNAs, thereby increasing synthesis of DNA damage repair proteins under conditions of increased turnover. Consequently, XPO1 inhibition decreased the capacity of lymphoma cells to repair DNA damage and ultimately resulted in increased cytotoxicity. In a phase I clinical trial conducted in R/R DLBCL, the combination of selinexor with second-line chemoimmunotherapy was tolerated with early indication of efficacy. Overall, this study reveals that XPO1 overexpression plays a critical role in the increased tolerance of cancer cells to DNA damage while providing new insights to optimize the clinical development of XPO1 inhibitors.
Significance: XPO1 regulates the dynamic ribonucleoprotein nuclear export in response to genotoxic stress to support tolerance and can be targeted to enhance the sensitivity of cancer cells to endogenous and exogenous DNA damage.
A related commentary is available here.
Journal: Cancer Research (January 15 issue)
Myelodysplastic syndromes with ring sideroblasts (MDS-RS) commonly develop from hematopoietic stem cells (HSC) bearing mutations in the splicing factor SF3B1 (SF3B1mt). Direct studies into MDS-RS pathobiology have been limited by a lack of model systems that fully recapitulate erythroid biology and RS development and the inability to isolate viable human RS. Here, we combined successful direct RS isolation from patient samples, high-throughput multiomics analysis of cells encompassing the SF3B1mt stem-erythroid continuum, and functional assays to investigate the impact of SF3B1mt on erythropoiesis and RS accumulation. The isolated RS differentiated, egressed into the blood, escaped traditional nonsense-mediated decay (NMD) mechanisms, and leveraged stress-survival pathways that hinder wild-type hematopoiesis through pathogenic GDF15 overexpression. Importantly, RS constituted a contaminant of magnetically enriched CD34+ cells, skewing bulk transcriptomic data. Mis-splicing in SF3B1mt cells was intensified by erythroid differentiation through accelerated RNA splicing and decreased NMD activity, and SF3B1mt led to truncations in several MDS-implicated genes. Finally, RNA mis-splicing induced an uncoupling of RNA and protein expression, leading to critical abnormalities in proapoptotic p53 pathway genes. Overall, this characterization of erythropoiesis in SF3B1mt RS provides a resource for studying MDS-RS and uncovers insights into the unexpectedly active biology of the “dead-end” RS.
Significance: Ring sideroblast isolation combined with state-of-the-art multiomics identifies survival mechanisms underlying SF3B1-mutant erythropoiesis and establishes an active role for erythroid differentiation and ring sideroblasts themselves in SF3B1-mutant myelodysplastic syndrome pathogenesis.
Journal: Clinical Cancer Research (January 1 issue)
Purpose: The monarcHER trial has shown that abemaciclib, a cyclin-dependent kinase 4 and 6 inhibitor, combined with fulvestrant and trastuzumab, improves progression-free survival (PFS) in hormone receptor–positive (HR+), HER2-positive (HER2+) advanced breast cancer (ABC) compared with standard-of-care (SOC) chemotherapy combined with trastuzumab. We report the final overall survival (OS) analysis, updated safety and efficacy data, and exploratory biomarker results from monarcHER.
Patients and Methods: monarcHER (NCT02675231), a randomized, multicenter, open-label, phase II trial, enrolled 237 patients across Arm A (abemaciclib, trastuzumab, fulvestrant), Arm B (abemaciclib, trastuzumab), and Arm C (SOC chemotherapy, trastuzumab). Following the statistical plan, OS and PFS were estimated in all arms. RNA sequencing (RNA-seq) was performed on archival tissue.
Results: Median OS was 31.1 months in Arm A, 29.2 months in Arm B, and 20.7 months in Arm C [A vs. C: HR, 0.71; 95% confidence interval (CI), 0.48–1.05; nominal two-sided P value 0.086; B vs. C: HR 0.83 (95% CI, 0.57–1.23); nominal two-sided P value 0.365]. Updated PFS and safety findings were consistent with previous results. The most frequently reported treatment-emergent adverse events included diarrhea, fatigue, nausea, neutrophil count decrease, and anemia. In exploratory RNA-seq analyses, Luminal subtypes were associated with longer PFS [8.6 vs. 5.4 months (HR, 0.54; 95% CI, 0.38–0.79)] and OS [31.7 vs. 19.7 months (HR, 0.68; 95% CI, 0.46–1.00)] compared with non-Luminal.
Conclusions: In this phase II trial, abemaciclib + trastuzumab ± fulvestrant numerically improved median OS in women with HR+, HER2+ ABC compared with SOC chemotherapy + trastuzumab.
This study was highlighted in the January 1 issue.
Journal: Clinical Cancer Research (January 15 issue)
Purpose: Hematopoietic cell transplantation (HCT) has curative potential for myeloid malignancies, though many patients cannot tolerate myeloablative conditioning with high-dose chemotherapy alone or with total-body irradiation (TBI). Here we report long-term outcomes from a phase I/II study using iodine-131 (131I)-anti-CD45 antibody BC8 combined with nonmyeloablative conditioning prior to HLA-haploidentical HCT in adults with high-risk relapsed/ refractory acute myeloid or lymphoid leukemia (AML or ALL), or myelodysplastic syndrome (MDS; ClinicalTrials.gov, NCT00589316).
Patients and Methods: Patients received a tracer diagnostic dose before a therapeutic infusion of 131Ianti-CD45 to deliver escalating doses (12–26 Gy) to the dose-limiting organ. Patients subsequently received fludarabine, cyclophosphamide (CY), and 2 Gy TBI conditioning before haploidentical marrow HCT. GVHD prophylaxis was posttransplant CY plus tacrolimus and mycophenolate mofetil.
Results: Twenty-five patients (20 with AML, 4 ALL and 1 high-risk MDS) were treated; 8 had ≥ 5% blasts by morphology (range 9%–20%), and 7 had previously failed HCT. All 25 patients achieved a morphologic remission 28 days after HCT, with only 2 patients showing minimal residual disease (0.002–1.8%) by flow cytometry. Median time to engraftment was 15 days for neutrophils and 23 days for platelets. Point estimates for overall survival and progression-free survival were 40% and 32% at 1 year, and 24% at 2 years, respectively. Point estimates of relapse and nonrelapse mortality at 1 year were 56% and 12%, respectively.
Conclusions: 131I-anti-CD45 radioimmunotherapy prior to haploidentical HCT is feasible and can be curative in some patients, including those with disease, without additional toxicity.
This study was highlighted in the January 15 issue.
Journal: Molecular Cancer Research

Metabolic Hallmarks for Purine Nucleotide Biosynthesis in Small Cell Lung Carcinoma
Small cell lung cancer (SCLC) has a poor prognosis, emphasizing the necessity for developing new therapies. The de novo synthesis pathway of purine nucleotides, which is involved in the malignant growth of SCLC, has emerged as a novel therapeutic target. Purine nucleotides are supplied by two pathways: de novo and salvage. However, the role of the salvage pathway in SCLC and the differences in utilization and crosstalk between the two pathways remain largely unclear. Here, we found that deletion of the HPRT1 gene, which codes for the rate-limiting enzyme of the purine salvage pathway, significantly suppressed tumor growth in vivo in several SCLC cells. We also demonstrated that HPRT1 expression confers resistance to lemetrexol (LMX), an inhibitor of the purine de novo pathway. Interestingly, HPRT1-knockout had less effect on SCLC SBC-5 cells, which are more sensitive to LMX than other SCLC cell lines, suggesting that a preference for either the purine de novo or salvage pathway occurs in SCLC. Furthermore, metabolome analysis of HPRT1-knockout cells revealed increased intermediates in the pentose phosphate pathway and elevated metabolic flux in the purine de novo pathway, indicating compensated metabolism between the de novo and salvage pathways in purine nucleotide biosynthesis. These results suggest that HPRT1 has therapeutic implications in SCLC and provide fundamental insights into the regulation of purine nucleotide biosynthesis.
Implications: SCLC tumors preferentially utilize either the de novo or salvage pathway in purine nucleotide biosynthesis, and HPRT1 has therapeutic implications in SCLC.
This article was highlighted and featured on the cover of the January issue.
Journal: Molecular Cancer Therapeutics
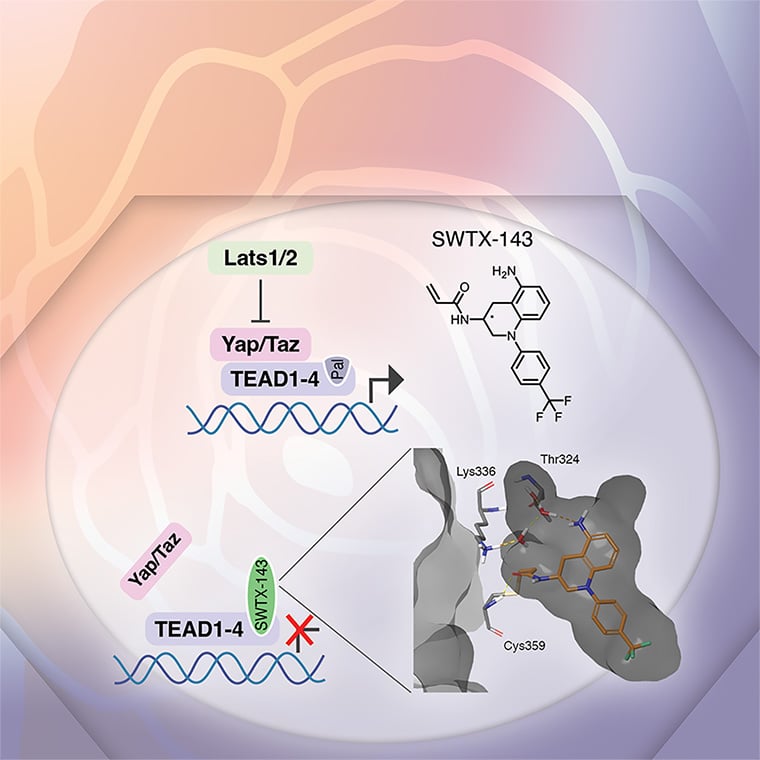
The Hippo pathway and its downstream effectors, the YAP and TAZ transcriptional coactivators, are deregulated in multiple different types of human cancer and are required for cancer cell phenotypes in vitro and in vivo, while largely dispensable for tissue homeostasis in adult mice. YAP/TAZ and their main partner transcription factors, the TEAD1–4 factors, are therefore promising anticancer targets. Because of frequent YAP/TAZ hyperactivation caused by mutations in the Hippo pathway components NF2 and LATS2, mesothelioma is one of the prime cancer types predicted to be responsive to YAP/TAZ-TEAD inhibitor treatment. Mesothelioma is a devastating disease for which currently no effective treatment options exist. Here, we describe a novel covalent YAP/TAZ-TEAD inhibitor, SWTX-143, that binds to the palmitoylation pocket of all four TEAD isoforms. SWTX-143 caused irreversible and specific inhibition of the transcriptional activity of YAP/TAZ-TEAD in Hippo-mutant tumor cell lines. More importantly, YAP/TAZ-TEAD inhibitor treatment caused strong mesothelioma regression in subcutaneous xenograft models with human cells and in an orthotopic mesothelioma mouse model. Finally, SWTX-143 also selectively impaired the growth of NF2-mutant kidney cancer cell lines, suggesting that the sensitivity of mesothelioma models to these YAP/TAZ-TEAD inhibitors can be extended to other tumor types with aberrations in Hippo signaling. In brief, we describe a novel and specific YAP/TAZ-TEAD inhibitor that has potential to treat multiple Hippo-mutant solid tumor types.
This study was highlighted and featured on the cover of the January issue.
Journal: Cancer Research Communications
Immune checkpoint inhibitors improve survival in patients with mismatch repair deficiency/microsatellite instability-high (MSI-H) colorectal cancer. The recurrence outcomes following discontinuation of immunotherapy after prolonged disease control have not been definitively reported in large series. Records from patients with advanced MSI-H colorectal cancer from The University of Texas – MD Anderson Cancer Center who received immunotherapy between 2014 and 2022 and stopped after prolonged clinical benefit were reviewed. Median progression-free and overall survival were estimated. Associations between the event of recurrence and coexisting mutations (KRAS/NRAS, BRAFV600E), metastatic organ involvement (lung, liver, lymph node, or peritoneum), metastatic timing (synchronous vs. metachronous), prior immunotherapy [anti-PD-(L)1 alone or in combination with anti-CTLA antibodies], etiology of MSI status (sporadic vs. hereditary non-polyposis colorectal cancer), and duration of immunotherapy were assessed. Sixty-four patients with MSI-H colorectal cancer without progression on immunotherapy were reviewed. Of these 48 and 16 received anti-PD(L)1 antibody alone or in combination with anti-CTLA-4 antibody, respectively. Median exposure to immunotherapy was 17.6 months (range, 1.3–51.9). After a median follow-up of 22.6 months (range, 0.3–71.7) after stopping immunotherapy, 56 of 64 patients (88%) remained without disease progression. Lung metastases were associated with recurrence/progression (OR, 6.1; P = 0.04), but coexisting mutation, primary tumor sidedness, and immunotherapy were not. These data provide a retrospective, single-institution analysis that showed that most patients with advanced MSI-H colorectal cancer do not recur after treatment cessation, regardless of the reason for stopping treatment or a variety of patient and disease features, supporting an optimistic prognosis of sustained disease control.
Significance: Outcomes for patients with MSI-H colorectal cancer stopping immunotherapy after disease control remain unknown. Sixty-four patients with MSI-H colorectal cancer from our institution stopping treatment for sustained benefit or toxicity were retrospectively assessed. After median follow up of 22 months and median immunotherapy exposure of 18 months, 88% patients remained without progression. All patients who recurred or progressed and were rechallenged with immunotherapy have continued to experience disease control.
An AACR press release on this study is available here.
Source link
#January #Highlights #AACR #Journals